Kazakhstan develops homegrown drug for mugwort pollen allergy
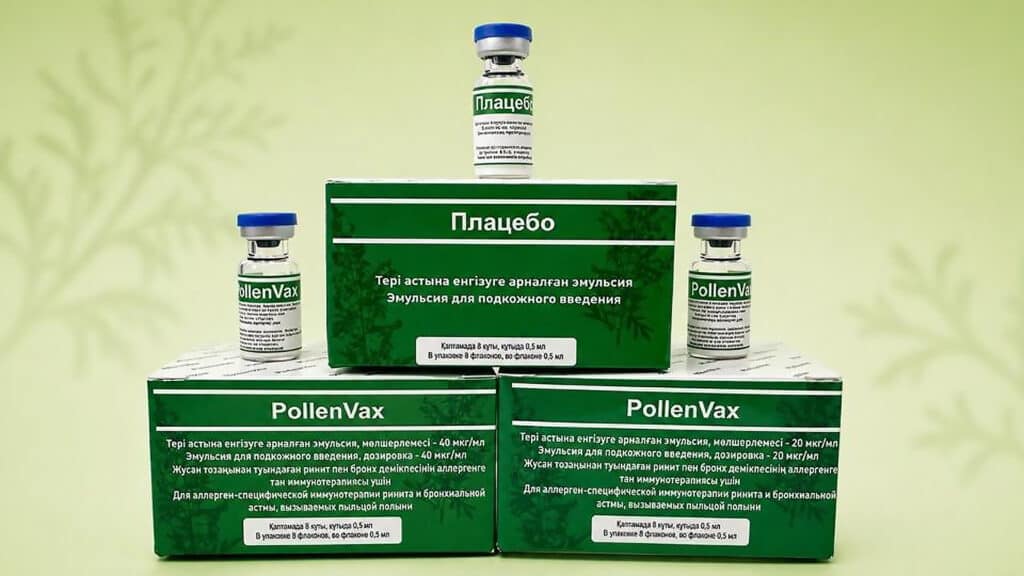
Scientists at the Kazakh National Agrarian Research University have begun the second phase of clinical trials of a drug to treat mugwort pollen allergy, the Ministry of Agriculture of Kazakhstan reported.
According to the ministry, the PollenVax immunotherapy drug, developed under the Ministry of Healthcare program, has successfully completed the first phase of trials assessing safety and tolerability. The next clinical trial will involve 138 volunteers and last 180 days.
«The drug is developed on the basis of a recombinant mugwort pollen allergen protein and is intended to provide a significantly shortened course of therapy—just four subcutaneous injections administered one week apart,» the statement said.
Previously, the first trial phase was successfully conducted involving 30 volunteers. According to the scientists, the drug demonstrated a favorable safety profile and tolerability. Furthermore, positive immunological results were reported, including the formation of allergen-blocking antibodies and reduced allergen sensitivity based on skin prick test results.
Mugwort pollen allergy is among the most widespread forms of seasonal allergy in Kazakhstan. According to local clinical diagnostic laboratories, sensitization to mugwort pollen ranks first among all identified pollen allergens, accounting for about 30% of cases. The issue is particularly relevant in Almaty, where the mugwort pollen season lasts from July to November. During this period, thousands of people experience symptoms of allergic rhinitis, including nasal congestion, sneezing, watery eyes, and reduced quality of life.

